Signal transduction in light–oxygen–voltage receptors lacking the adduct-forming cysteine residue
Estella F. Yee, Ralph P. Diensthuber, Anand T. Vaidya, Peter P. Borbat, Christopher Engelhard, Jack H. Freed, Robert Bittl, Andreas Möglich & Brian R. Crane
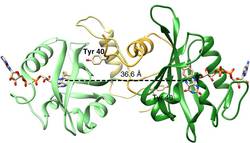
Light–oxygen–voltage (LOV) receptors sense blue light through the photochemical generation of a covalent adduct between a flavin-nucleotide chromophore and a strictly conserved cysteine residue. Here we show that, after cysteine removal, the circadian-clock LOV-protein Vivid still undergoes light-induced dimerization and signalling because of flavin photo-reduction to the neutral semiquinone (NSQ). Similarly, photoreduction of the engineered LOV histidine kinase YF1 to the NSQ modulates activity and downstream effects on gene expression.
Nature Communications 2015, | 6:10079 | DOI: 10.1038/nchembio.1890
Orientation-Controlled Electrocatalytic Efficiency of an Adsorbed Oxygen-Tolerant Hydrogenase
N. Heidary, T. Utesch, M. Zerball, M. Horch, D. Millo, J. Fritsch, O. Lenz, R. von Klitzing, P. Hildebrandt, A. Fischer, M. A. Mroginski, I. Zebger
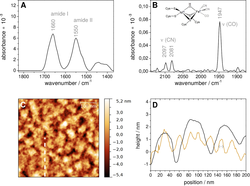
Protein immobilization on electrodes is a key concept in exploiting enzymatic processes for bioelectronic devices. For optimum performance, an in-depth understanding of the enzyme-surface interactions is required. Here, we introduce an integral approach of experimental and theoretical methods that provides detailed insights into the adsorption of an oxygen-tolerant [NiFe] hydrogenase on a biocompatible gold electrode.
Using atomic force microscopy, ellipsometry, surface-enhanced IR spectroscopy, and protein film voltammetry, we explore enzyme coverage, integrity, and activity, thereby probing both structure and catalytic H2 conversion of the enzyme. Electrocatalytic efficiencies can be correlated with the mode of protein adsorption on the electrode as estimated theoretically by molecular dynamics simulations. Our results reveal that pre-activation at low potentials results in increased current densities, which can be rationalized in terms of a potential-induced re-orientation of the immobilized enzyme.
PLOS ONE 2015, | DOI: 10.1371/journal.pone.0143101
Concentric-flow electrokinetic injector enables serial crystallography of ribosome and photosystem II
Raymond G Sierra, Cornelius Gati, Hartawan Laksmono, E Han Dao, Sheraz Gul, Franklin Fuller, Jan Kern, Ruchira Chatterjee, Mohamed Ibrahim, Aaron S Brewster, Iris D Young, Tara Michels-Clark, Andrew Aquila, Mengning Liang, Mark S Hunter, Jason E Koglin, Sébastien Boutet, Elia A Junco, Brandon Hayes, Michael J Bogan, Christina Y Hampton, Elisabetta V Puglisi, Nicholas K Sauter, Claudiu A Stan, Athina Zouni, Junko Yano, Vittal K Yachandra, S Michael Soltis, Joseph D Puglisi & Hasan DeMirci
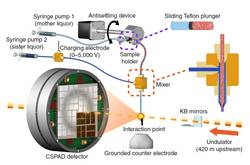
We describe a concentric-flow electrokinetic injector for efficiently delivering microcrystals for serial femtosecond X-ray crystallography analysis that enables studies of challenging biological systems in their unadulterated mother liquor. We used the injector to analyze microcrystals of Geobacillus stearothermophilus thermolysin (2.2-Å structure), Thermosynechococcus elongatus photosystem II (<3-Å diffraction) and Thermus thermophilus small ribosomal subunit bound to the antibiotic paromomycin at ambient temperature (3.4-Å structure).
Nature Methods 2015 | DOI: 10.1038/nmeth.3667
Minimum Information about a Biosynthetic Gene cluster
Marnix H. Medema, Daniel Petras, Roderich Süssmuth and coworkers
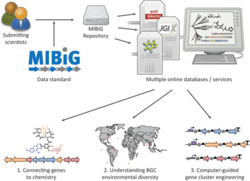
A wide variety of enzymatic pathways that produce specialized metabolites in bacteria, fungi and plants are known to be encoded in biosynthetic gene clusters. Information about these clusters, pathways and metabolites is currently dispersed throughout the literature, making it difficult to exploit.
To facilitate consistent and systematic deposition and retrieval of data on biosynthetic gene clusters, we propose the Minimum Information about a Biosynthetic Gene cluster (MIBiG) data standard.
Nature Chemical Biology 2015, 11, 625–631 | DOI: 10.1038/nchembio.1890
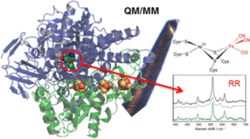
Resonance Raman Spectroscopic Analysis of the [NiFe] Active Site and the Proximal [4Fe-3S] Cluster of an O2-Tolerant Membrane-Bound Hydrogenase in the Crystalline State
E. Siebert, Y. Rippers, S. Frielingsdorf, J. Fritsch, A. Schmidt, J. Kalms, S. Katz, O. Lenz, P. Scheerer, L. Paasche, V. Pelmenschikov, U. Kuhlmann, M. A. Mroginski, I. Zebger, P. Hildebrandt
We have applied resonance Raman (RR) spectroscopy on single protein crystals of the O2-tolerant membrane-bound [NiFe] hydrogenase (MBH from Ralstonia eutropha) which catalyzes the splitting of H2 into protons and electrons.
RR spectra taken from 65 MBH samples in different redox states were analyzed in terms of the respective component spectra of the active site and the unprecedented proximal [4Fe-3S] cluster using a combination of statistical methods and global fitting procedures. These component spectra of the individual cofactors were compared with calculated spectra obtained by quantum mechanics/molecular mechanics (QM/MM) methods.
Thus, the recently discovered hydroxyl-coordination of one iron in the [4Fe-3S] cluster was confirmed. Infrared (IR) microscopy of oxidized MBH crystals revealed the [NiFe] active site to be in the Nir-B [Ni(III)] and Nir-S [Ni(II)] states, whereas RR measurements of these crystals uncovered the Nia-S [Ni(II)] state as the main spectral component, suggesting its in situ formation via photodissociation of the assumed bridging hydroxyl or water ligand.
On the basis of QM/MM calculations, individual band frequencies could be correlated with structural parameters for the Nia-S state as well as for the Ni-L state, which is formed upon photodissociation of the bridging hydride of H2-reduced active site states.
J. Phys. Chem. B 2015, 119, 13785–13796 | DOI: 10.1021/acs.jpcb.5b04119
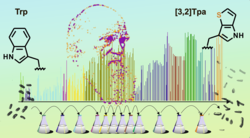
Chemical Evolution of a Bacterial Proteome
Michael Georg Hoesl, Stefan Oehm, Patrick Durkin, Elise Darmon, Lauri Peil, Hans-Rudolf Aerni, Juri Rappsilber, Jesse Rinehart, David Leach, Dieter Söll, and Nediljko Budisa
We have changed the amino acid set of the genetic code of Escherichia coli by evolving cultures capable of growing on the synthetic noncanonical amino acid L-β-(thieno[3,2-b]pyrrolyl)alanine ([3,2]Tpa) as a sole surrogate for the canonical amino acid L-tryptophan (Trp).
A long-term cultivation experiment in defined synthetic media resulted in the evolution of cells capable of surviving Trp![]() [3,2]Tpa substitutions in their proteomes in response to the 20 899 TGG codons of the E. coli W3110 genome.
[3,2]Tpa substitutions in their proteomes in response to the 20 899 TGG codons of the E. coli W3110 genome.
These evolved bacteria with new-to-nature amino acid composition showed robust growth in the complete absence of Trp. Our experimental results illustrate an approach for the evolution of synthetic cells with alternative biochemical building blocks.
Angew. Chem. Int. Ed. 2015, 54, 10030-34 | DOI: 10.1002/anie.201502868
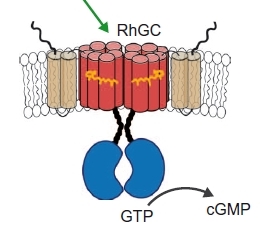
The rhodopsin–guanylyl cyclase of the aquatic fungus Blastocladiella emersonii enables fast optical control of cGMP signaling
Ulrike Scheib, Katja Stehfest, Christine E. Gee, Heinz G. Körschen, Roman Fudim,
Thomas G. Oertner, Peter Hegemann
The second messenger cyclic guanosine monophosphate (cGMP) alters ion channel activity to mediate processes such as smooth muscle relaxation, transduction of light, and apoptosis.
Guanylyl cyclases produce cGMP from GTP. Scheib et al. characterized the fungal photoreceptor RhGC, which consists of the light-sensitive receptor rhodopsin connected to a guanylyl cyclase.
When expressed in mammalian cells, including neurons, light stimulated RhGC to generate cGMP. Thus, RhGC is another tool in the optogenetic kit and can be used to study cGMP signaling.
Science Signaling 2015, 8, rs8 | DOI: 10.1126/scisignal.aab0611
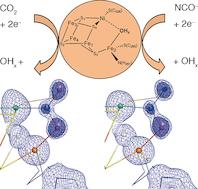
How the [NiFe4S4] Cluster of CO Dehydrogenase Activates Carbon Dioxide and NCO−
Jens Jochen Fesseler, Jae-Hun Jeoung and Holger Dobbek
Ni,Fe-containing CO dehydrogenases (CODHs) use a [NiFe4S4] cluster, termed cluster C, to reversibly reduce CO2 to CO with high turnover number. Binding to Ni and Fe activates CO2, but current crystal structures have insufficient resolution to analyze the geometry of bound CO2 and reveal the extent and nature of its activation. The crystal structures of CODH in complex with CO2 and the isoelectronic inhibitor NCO− are reported at true atomic resolution (dmin≤1.1 Å). Like CO2, NCO− is a μ2,η2 ligand of the cluster and acts as a mechanism-based inhibitor.
While bound CO2 has the geometry of a carboxylate group, NCO− is transformed into a carbamoyl group, thus indicating that both molecules undergo a formal two-electron reduction after binding and are stabilized by substantial π backbonding. The structures reveal the combination of stable μ2,η2 coordination by Ni and Fe2 with reductive activation as the basis for both the turnover of CO2 and inhibition by NCO−.
Angew. Chem. Int. Ed. 2015, 54, 1 - 6 | DOI: 10.1002/anie.201501778
Surface-Tuned Electron Transfer and Electrocatalysis of Hexameric Tyrosine-Coordinated Heme Protein
L. Peng, T. Utesch, A. Yarman, J. H. Jeoung, S. Steinborn, H. Dobbek, M. A. Mroginski, J. Tanne, U. Wollenberger, F. W. Scheller
Molecular modeling, electrochemical methods, and quartz crystal microbalance were used to characterize immobilized hexameric tyrosine-coordinated heme protein (HTHP) on bare carbon or on gold electrodes modified with positively and negatively charged self-assembled monolayers (SAMs), respectively.
HTHP binds to the positively charged surface but no direct electron transfer (DET) is found due to the long distance of the active sites from the electrode surfaces. At carboxyl-terminated surfaces, the neutrally charged bottom of HTHP can bind to the SAM. For this “disc” orientation all six hemes are close to the electrode and their direct electron transfer should be efficient. HTHP on all negatively charged SAMs showed a quasi-reversible redox behavior with rate constant ks values between 0.93 and 2.86 s−1 and apparent formal potentials between -131.1 and -249.1 mV. On the MUA/MU-modified electrode, the maximum surface concentration corresponds to a complete monolayer of the hexameric HTHP in the disc orientation. HTHP electrostatically immobilized on negatively charged SAMs shows electrocatalysis of peroxide reduction and enzymatic oxidation of NADH.
Chemistry A European Journal 2015, 21, 7596-7602| DOI: 10.1002/chem.201405932
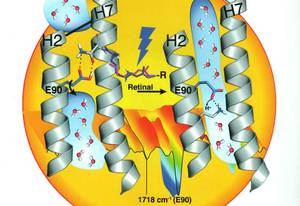
Early Formation of the Ion-Conducting Pore in Channelrhodopsin-2
Jens Kuhne, Kirstin Eisenhauer, Eglof Ritter, Peter Hegemann, Klaus Gerwert, and Franz Bartl
Channelrhodopsins (ChRs) are light-gated ion channels that are widely used in optogenetics. They allow precise control of neuronal activity with light, but a detailed understanding of how the channel is gated and the ions are conducted is still lacking.
The recent determination of the X-ray structural model in the closed state marks an important milestone. Herein the open state structure is presented and the early formation of the ion conducting pore is elucidated in atomic detail using time-resolved FTIR spectroscopy. Photoisomerization of the retinal-chromophore causes a downward movement of the highly conserved E90, which opens the pore. ...
Angew. Chem. Int. Ed. 2015, 54, 4953-4957 | DOI: 10.1002/anie.201410180
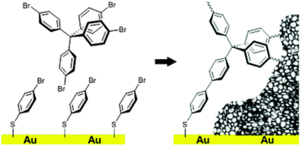
Microporous polymer network films covalently bound to gold electrodes
D. Becker, N. Heidary, M. Horch, U. Gernert, I. Zebger, J. Schmidt, A. Fischer, A. Thomas
Covalent attachment of a microporous polymer network (MPN) on a gold surface is presented. A functional bromophenyl-based self-assembled monolayer (SAM) formed on the gold surface acts as co-monomer in the polymerisation of the MPN yielding homogeneous and robust coatings. Covalent binding of the films to the electrode is confirmed by SEIRAS measurements.
Chemical Communications 2015, 51, 4283-4286| DOI: 10.1039/C4CC09637A
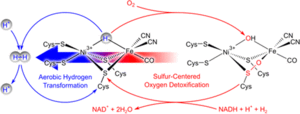
Reversible Active Site Sulfoxygenation Can Explain the Oxygen Tolerance of a NAD+-Reducing [NiFe] Hydrogenase and Its Unusual Infrared Spectroscopic Properties
Marius Horch, Lars Lauterbach, Maria Andrea Mroginski, Oliver Lenz, Peter Hildebrandt, and Ingo Zebger
Oxygen-tolerant [NiFe] hydrogenases are metalloenzymes that represent valuable model systems for sustainable H2 oxidation and production. The soluble NAD+-reducing [NiFe] hydrogenase (SH) from Ralstonia eutropha couples the reversible cleavage of H2 with the reduction of NAD+ and displays a unique O2 tolerance.
Here we performed IR spectroscopic investigations on purified SH in various redox states in combination with density functional theory to provide structural insights into the catalytic [NiFe] center. These studies revealed a standard-like coordination of the active site with diatomic CO and cyanide ligands.
The long-lasting discrepancy between spectroscopic data obtained in vitro and in vivo could be solved on the basis of reversible cysteine oxygenation in the fully oxidized state of the [NiFe] site. The data are consistent with a model in which the SH detoxifies O2 catalytically by means of an NADH-dependent (per)oxidase reaction involving the intermediary formation of stable cysteine sulfenates. The occurrence of two catalytic activities, hydrogen conversion and oxygen reduction, at the same cofactor may inspire the design of novel biomimetic catalysts performing H2-conversion even in the presence of O2.
J. Am. Chem. Soc. 2015, 137, 2555-2564 | DOI: 10.1021/ja511154y