News
Redox-dependent substrate-cofactor interactions in the Michaelis-complex of a flavin-dependent oxidoreductase
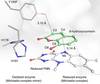
New research highlight: Peter Hildebrandt, Holger Dobbek, Ingo Zebger, and coworkers investigate the Michaelis complex of the flavoenzyme xenobiotic reductase A with the reactive reduced cofactor bound to its substrates by X-ray crystallography and resonance Raman spectroscopy and compare it to the non-reactive oxidized Michaelis complex mimics.
A Decade of Excellent Catalysis Research

The 10th anniversary of their existence is being celebrated with the symposium entitled “Unifying Concepts in Catalysis: Status Quo and Challenges” at the TU Berlin.
10th Anniversary of UniCat: Symposium on "Unifying Concepts in Catalysis: Status Quo and Challenges."

The Cluster of Excellence, UniCat, and its graduate school, the BIG-NSE are going to celebrate their 10th anniversary at the TU Berlin from July 12th – 14th.
The students from my BIG-NSE year still meet once a week, and we regularly discuss our work

To mark the 10-year anniversary of BIG-NSE, UniCat asks current members of the graduate school about their experiences and impressions
Insights into trans-Ligand and Spin-Orbit Effects on Electronic Structure and Ligand NMR Shifts in Transition-Metal Complexes

Invited for the cover of this issue is the group of Martin Kaupp at the Technical University Berlin. The image depicts remark-ably general and large trans ligand effects on NMR shifts in 5d transition-metal complexes, and their main MO origins.
It made me feel unique to synthesize something which didn’t exist before

Interview with current Berlin International Graduate School of Natural Sciences and Engineering (BIG-NSE) fellow Min Ha Kim on the occasion of the 10th Anniversary of BIG-NSE
Harvey Prize in Human Health for Peter Hegemann

The 2016 Harvey Prize in the field of Human Health was awarded to Prof. Peter Hegemann and Prof. Karl Diesseroth on 11 June 2017 at the Technion Institute of Technology in Haifa, Israel.
Research Highlight "Reversible light-dependent molecular switches on Ag/AgCl nanostructures"
Nanostructured Ag/AgCl substrates were used to generate reversible and highly efficient light-dependent chemical switches based on adsorbed 4,4’-dimercaptoazobenzene (DMAB). DMAB was formed in situ via laser-induced dimerization either from 4-nitrothiophenol (4-NTP) or 4-aminothiophenol (4-ATP). The subsequent reaction pathways of DMAB, however, were quite different as monitored by surface enhanced Raman spectroscopy.
Carbon Monoxide Dehydrogenase Reduces Cyanate to Cyanide
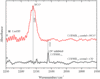
The biocatalytic function of carbon monoxide dehydrogenase (CODH) has a high environmental relevance owing to its ability to reduce CO2. Despite numerous studies on CODH over the past decades, its catalytic mechanism is not yet fully understood. In the present combined spectroscopic and theoretical study, we report first evidences for a cyanate (NCO-) to cyanide (CN-) reduction at the C-cluster.

