Towards biotechnological application of oxygen-tolerant hydrogenases
The mechanistic understanding of biosynthesis and catalysis of O2-tolerant hydrogenases in conjunction with the possibility of obtained large amounts of highly purified enzyme provides the basis for new potential applications of these complex biocatalysts.
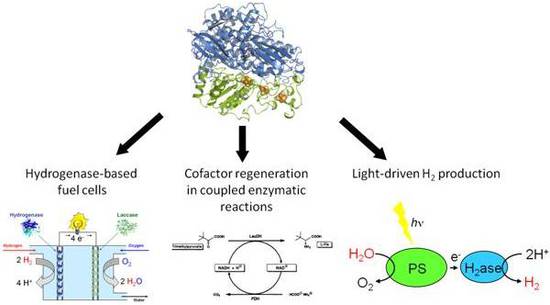
Because of their ability to catalyse both H2 consumption and H2 production under aerobic conditions, O2-tolerant hydrogenases have received great attention as attractive catalysts for biotechnological application.
This includes enzymatic fuel cells as well as light driven H2-production by hybrid protein complexes consisting of a hydrogenase directly coupled to PSI. By exploiting natural protein-protein interactions, we have obtained the highest H2 production rates for comparable in vitro systems.
Research goals
- Central objectives are the design of durable and efficient hydrogenase-based biofuel cells and the development of in vivo devices for light-driven hydrogen formation.
- In order to wire hydrogenase to PSI for light-driven H2 production, established and new coupling strategies will be employed, including direct and indirect coupling via natural protein-protein interactions and electron mediators (e.g. ferredoxin, NADPH), respectively.
- Biofuel cells based on electrodes of nanoporous materials with pore sizes of tens of nanometers and appropriate surface functionalisation will be developed and used for immobilising hydrogenases with a high load in order to improve the capacity and long-term stability of hydrogenase- based biofuel cells.
- We will genetically engineer R. eutropha cells in order to introduce conducting wires. The engineered cells may be embedded into stabilizing conducting nanomaterials, powered for instance by solar energy.
- Engineered cellular systems will be extended to generate H2/CO/CO2-based microbial factories for biofuel production (see D1-2).