Active Mixed-Valent MnOx Water Oxidation Catalysts through Partial Oxidation of Nanostructured MnO Particles
Arindam Indra, Prashanth W. Menezes, Ivelina Zaharieva, Elham Baktash, Johannes Pfrommer, Michael Schwarze, Holger Dau,* and Matthias Driess *
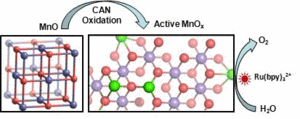
The unexpected facile conversion of catalytically inactive nanostructured crystalline manganese monoxide (MnO) into photo- and electrocatalytically active amorphous MnOx catalysts for effective water oxidation using CeIV ions as an oxidant has been described for the first time.
Extended X-ray absorption fine structure (EXAFS) analyses of the active MnOx catalyst reveals the presence of MnII, MnIII, and MnIV centers interconnected through oxido bridges. The structural motif of the active MnOx is close to biomimetic photosystem II catalysts; however, the catalyst does not need the presence of calcium ions to show efficient catalytic activity in photo- and electrochemical water oxidation.
This paper has been selected as a very important paper in Angewandte Chemie.
Angew. Chem. Int. Ed. 2013; DOI: anie.201307543
Published online 31 October 2013.
Access to a CuII–O–CuII Motif: Spectroscopic Properties, Solution Structure, and Reactivity
Peter Haack, Anne Kärgel, Claudio Greco, Jadranka Dokic, Beatrice Braun, Florian Felix Pfaff, Stefan Mebs, Kallol Ray, Christian Limberg*
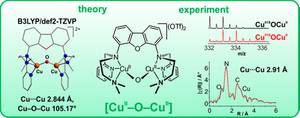
The groups of Ray and Limberg report a complex with a rare CuII–O–CuII structural motif that is stable at room temperature, which allows its in-depth characterization by a variety of spectroscopic methods. Interest in such compounds is fueled by the recent discovery that a CuII–O–CuII species on the surface of Cu-ZSM-5 is capable of oxidizing methane to methanol, and this in turn ties into mechanistic discussions on the methane oxidation at the dicopper site within the particulate methane monooxygenase.
For the synthesis of the Cu2O complex Limberg et al. have developed a novel, neutral ligand system, FurNeu, exhibiting two N-(N′,N′-dimethylaminoethyl)(2-pyridylmethyl)amino binding pockets connected by a dibenzofuran spacer. ...
J. Am. Chem. Soc. 2013, 135, 16148 – 16160.
Published online 17 October 2013.
Methane Coupling over Magnesium Oxide: How Doping Can Work
Pierre Schwach, Marc Georg Willinger, Annette Trunschke, and Robert Schlögl
In the present work, we put the concept to test with powder catalysts working at high temperature (T=1023 K). We synthesized doped magnesium oxide as it has been investigated frequently in OCM. Pure magnesium oxide is deactivated at this temperature quite rapidly and reaches a stationary state after a few minutes to several hours depending on the applied contact time and the initial nanostructure of the magnesium oxide. …
We introduced Fe in ppm quantities into MgO. The synthesis of Fe-doped polycrystalline magnesium oxide in which the Fe dopant is homogeneously distributed over the entire bulk is, however, quite challenging and requires highly sensitive analytical techniques for verification. The issue is distributing the dopant throughout the bulk of MgO in such a way that no precipitates or segregated nanostructured dopant phases occur under the drastic reaction conditions.
Angew. Chem. Int. Ed. 2013, 52, 11381 - 11384
Published online 17 September 2013.
Dedicated to Professor Helmut Schwarz on the occasion of his 70th birthday.
Coupling Biocatalysis with Molecular Imprinting in a Biomimetic Sensor
Aysu Yarman and Frieder W. Scheller
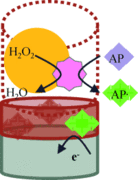
Make it simple: A molecularly imprinted electropolymer was combined with an enzyme in a catalytic biomimetic sensor that enabled interference-free detection of the drug aminopyrine (AP) at submicromolar concentrations in the presence of ascorbic acid and uric acid within 15 s. The sensor functioned by the peroxide-dependent conversion of AP in a layer above a product-imprinted electropolymer on an indicator electrode (see picture)
Angew. Chem. Int. Ed. 2013, 52, 11521-11525
Published online 09 September 2013.
Thermal Methane Activation by a Binary V–Nb Transition-Metal Oxide Cluster Cation: A Further Example for the Crucial Role of Oxygen-Centered Radicals
Zhe-Chen Wang, Jian-Wen Liu, Maria Schlangen, Thomas Weiske, Detlef Schröder, Joachim Sauer, and Helmut Schwarz
While often being considered as one of the “holy grails” in chemistry, the selective activation and functionalization of hydrocarbons, especially methane, encounter quite a few obstacles. For example, as the most stable alkane, methane possesses the strongest C-H bond of any saturated hydrocarbon. Furthermore, the negative electron affinity, small polarizability, large ionization energy, wide HOMO/LUMO gap, and an extremely high pKa value are also intrinsic features which make methane activation rather difficult.
Within this highlighted paper Sauer and Schwarz report on the heteronuclear transition-metal oxide cluster, that activates methane: VNbO5+ reacts with CH4 under ambient conditions via hydrogen-atom transfer (HAT), thus providing an interesting prototype example of room-temperature methane activation by a binary transition-metal oxide cluster.
Chem. Eur. J. 2013, 19, 11496–11501.
Published online 09 August 2013.
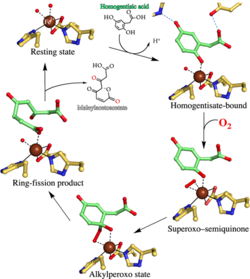
Visualizing the substrate-, superoxo-, alkylperoxo-, and product-bound states at the nonheme Fe(II) site of homogentisate dioxygenase
Jae-Hun Jeoung, Martin Bommer, Tzong-Yuan Lin, and Holger Dobbek
Homogentisate 1,2-dioxygenase (HGDO) uses a mononuclear nonheme Fe2+ to catalyze the oxidative ring cleavage in the degradation of Tyr and Phe by producing maleylacetoacetate from homogentisate (2,5-dihydroxyphenylacetate). Here, we report three crystal structures of HGDO, revealing five different steps in its reaction cycle at 1.7–1.98 Å resolution. The resting state structure displays an octahedral coordination for Fe2+ with two histidine residues (His331 and His367), a bidentate carboxylate ligand (Glu337), and two water molecules.
Homogentisate binds as a monodentate ligand to Fe2+, and its interaction with Tyr346 invokes the folding of a loop over the active site, effectively shielding it from solvent. Binding of homogentisate is driven by enthalpy and is entropically disfavored as shown by anoxic isothermal titration calorimetry. Three different reaction cycle intermediates have been trapped in different HGDO subunits of a single crystal showing the influence of crystal packing interactions on the course of enzymatic reactions. ...
PNAS 2013; doi: 10.1073/pnas.1302144110
Published online 15 July 2013.
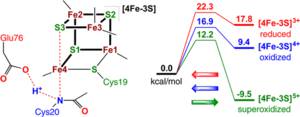
Redox-Dependent Structural Transformations of the [4Fe-3S] Proximal Cluster in O2-Tolerant Membrane-Bound [NiFe]-Hydrogenase: A DFT Study
Vladimir Pelmenschikov and Martin Kaupp
Broken-symmetry density functional theory (BS-DFT) has been used to address the redox-dependent structural changes of the proximal [4Fe-3S] cluster, implicated in the O2-tolerance of membrane-bound [NiFe]-hydrogenase (MBH).
The recently determined structures of the [4Fe-3S] cluster together with its protein ligands were studied at the reduced [4Fe-3S]3+, oxidized [4Fe-3S]4+, and superoxidized [4Fe-3S]5+ levels in context of their relative energies and protonation states. The observed proximal cluster conformational switch, concomitant with the proton transfer from the cysteine Cys20 backbone amide to the nearby glutamate Glu76 carboxylate, is found to be a single-step process requiring ~12–17 kcal/mol activation energy at the superoxidized [4Fe-3S]5+ level. At the more reduced [4Fe-3S]4+/3+ oxidation levels, this rearrangement has at least 5 kcal/mol higher activation barriers and prohibitively unfavorable product energies.
The reverse transformation of the proximal cluster is a fast unidirectional process with ~ 8 kcal/mol activation energy, triggered by one-electron reduction of the superoxidized species. A previously discussed ambiguity of the Glu76 carboxylate and ‘special’ Fe4 iron positions in the superoxidized cluster is now rationalized as a superposition of two local minima, where Glu76-Fe4 coordination is either present or absent. The calculated 12.3–17.9 MHz 14N hyperfine coupling (HFC) for the Fe4-bound Cys20 backbone nitrogen is in good agreement with the large 13.0/14.6 MHz 14N couplings from the latest HYSCORE/ENDOR studies.
J. Am. Chem. Soc. 2013, 135, 11809 – 11823.
Published online 13 July 2013.
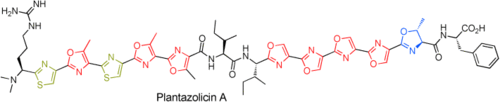
Total Synthesis of the Ribosomally Synthesized Linear Azole-Containing Peptide Plantazolicin A from Bacillus amyloliquefaciens
Dr. Srinivas Banala, Paul Ensle, and Roderich D. Süssmuth
A cyclodehydration reaction has been used to synthesize the linear azole-containing peptide plantazolicin A. The target compound was synthesized from two heterocyclic fragments derived from dipeptide building blocks. The acid-labile oxazolines and thiazoles necessitate the use of a Teoc/TMSE-based protection-group strategy, which allows structure modification of plantazolicin.
Angew. Chem. Int. Ed. 2013, 52, 9518 - 9523
Published online 11 June 2013.
Dedicated to the Bayer company on the occasion of its 150th anniversary.
Resonance Raman Spectroscopy as a Tool to Monitor the Active Site of Hydrogenases
Elisabeth Siebert, Marius Horch, Yvonne Rippers, Johannes Fritsch, Stefan Frielingsdorf, Oliver Lenz, Francisco Velazquez Escobar, Friedrich Siebert, Lars Paasche, Uwe Kuhlmann, Friedhelm Lendzian, Maria-Andrea Mroginski, Ingo Zebger, and Peter Hildebrandt
The biological conversion of hydrogen is catalyzed by [NiFe] hydrogenases utilizing a tailored bimetallic center. In their Communication Peter Hildebrandt, Ingo Zebger and co-workers use resonance Raman spectroscopy for the first time to characterize this active site by directly probing Fe-CO/CN vibrational modes. Applying an integrated spectroscopic and computational approach, this method provides new insights into structural and photochemical aspects of the [NiFe] site.
This paper has been selected as a very important paper in Angewandte Chemie and chosen as back cover.
Angew. Chem. Int. Ed. 2013, 19, 5162 - 5165
Published online 22 April 2013.
Synthesis and characterization of a high–valent mixed metal [CuIII(µ–O)2NiIII]2+ core with nucleophilic oxo groups
Subrata Kundu, Florian Felix Pfaff, Enrico Miceli, Ivelina Zaharieva, Christian Herwig, Shenglai Yao, Erik R. Farquhar, Uwe Kuhlmann, Eckhard Bill, Peter Hildebrandt, Holger Dau, Matthias Driess*, Christian Limberg*, Kallol Ray*
The groups of Ray, Driess, Limberg, Dau and Hildebrandt report the synthesis and spectroscopic characterization of a rare heterobimetallic high valent CuNi bis(µ–oxo) complex that unlike typical homodinuclear high–valent [M2(µ–O)2]n+ cores, behaves as nucleophiles, and can initiate deformylation of aldehydes. This report strongly suggests that mixed–metal bis(µ–oxo) cores are viable intermediates during the deformylation of fatty aldehydes by the enzyme cyanobacterial aldehyde decarbonylase, where a heterodinuclear cofactor has been suggested, but not yet isolated.
This paper has been selected as a very important paper in Angewandte Chemie.
Angew. Chem. Int. Ed. 2013, 52, 1 – 6.
Published online 15 April 2013.
Simultaneous Femtosecond X-ray Spectroscopy and Diffraction of Photosystem II at Room Temperature
Jan Kern, Roberto Alonso-Mori, Rosalie Tran, Johan Hattne, Richard J. Gildea, Nathaniel Echols, Carina Glöckner, Julia Hellmich, Hartawan Laksmono, Raymond G. Sierra, Benedikt Lassalle-Kaiser, Sergey Koroidov, Alyssa Lampe, Guangye Han, Sheraz Gul, Dörte DiFiore, Despina Milathianaki, Alan R. Fry, Alan Miahnahri, Donald W. Schafer, Marc Messerschmidt, M. Marvin Seibert, Jason E. Koglin, Dimosthenis Sokaras, Tsu-Chien Weng, Jonas Sellberg, Matthew J. Latimer, Ralf W. Grosse-Kunstleve, Petrus H. Zwart, William E. White, Pieter Glatzel, Paul D. Adams, Michael J. Bogan, Garth J. Williams, Sébastien Boutet, Johannes Messinger, Athina Zouni, Nicholas K. Sauter, Vittal K. Yachandra, Uwe Bergmann, Junko Yano
Science, published online on 14 February 2013
Intense femtosecond x-ray pulses produced at the Linac Coherent Light Source (LCLS) were used for simultaneous x-ray diffraction (XRD) and x-ray emission spectroscopy (XES) of microcrystals of Photosystem II (PS II) at room temperature.
This method probes the overall protein structure and the electronic structure of the Mn4CaO5 cluster in the oxygen-evolving complex of PS II. XRD data are presented from both the dark state (S1) and the first illuminated state (S2) of PS II. Our simultaneous XRD/XES study shows that the PS II crystals are intact during our measurements at the LCLS, not only with respect to the structure of PS II, but also with regard to the electronic structure of the highly radiation-sensitive Mn4CaO5 cluster, opening new directions for future dynamics studies.