Photosynthesis under the X-Ray
7th November 2014
New technology allows the analysis of biomolecules in a native-like form
Photosynthesis is among the most important processes of nature. The process through which all green plants harvest sunlight and thereby produce the oxygen in our air is, however, still not understood in detail today.
UniCat researchers Athina Zouni and Holger Dobbek have now illuminated a subsystem of photosynthesis in a native-like form with help from the X-ray light source, Petra III at the German Electron Synchrotron (DESY).
The X-ray examination of the so-called Photosystem II revealed, among other things, previously unknown structures, as reported by the scientists working with Adjunct Professor Dr. Athina Zouni and Prof. Dr. Holger Dobbek (both in UniCat Research Field E3 and from the HU Berlin) in the journal "Structure".
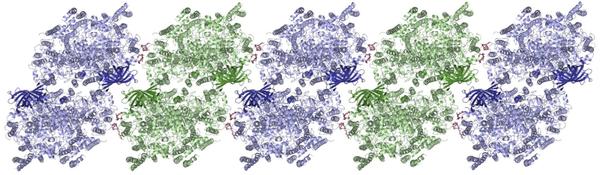
The molecular structure of the Photosystem II, which forms itself into rows in the cell membrane. Picture: Martin Bommer / HU Berlin
Photosystem II is the part of the photosynthetic machinery that divides water into hydrogen and oxygen with help of sunlight. It belongs to the membrane proteins. Membrane proteins are a large and important group of biomolecules that are of significance to, among other things, a number of medical questions. In order to decipher the structure of proteins, from which the details of the function of a biomolecule can be found, researchers use the very bright and short-wavelength X-radiation of PETRA III and similar systems. Small crystals must be grown from the biomolecules for this first.
Biomolecules - and in particular membrane proteins - usually only reluctantly allow themselves to be forced into crystal form, because it is contrary to their natural state. The preparation of suitable samples is therefore an art in and of itself. Thus, the Photosystem II must first be released from the membrane, where it is tied to numerous small fat molecules (lipids). For this, researchers use special detergents such as those found in soap. The catch: instead of by lipids, the biomolecules are now surrounded by detergents, which can make the crystal spongy, thus worsening the analysis. "What we want is as much nature as possible," says Zouni. Because the closer the proteins in the crystal are to their natural state, the better the results.
Zouni’s group has now succeeded in producing Photosystem II crystals, which no longer contain any detergents, so that nearly the native form of the biomolecules is frozen within them. "The trick was to use a detergent that is very different in composition and structure from the lipids," explains the Berlin researcher. Prior to the X-ray examination of these biomolecule crystals, part of their water is often removed and replaced by an antifreeze. Because the crystals are usually frozen for the study so that the high-energy X-rays do not harm them so quickly, the researchers want to avoid ice forming. "Dehydration has in our case not only withdrawn the water from our samples, but has also completely removed the detergent, that we did not expect," says Zouni. "What we have received through this, are samples very close to their native form, as no one had ever imagined before."
The examination revealed structures that were not visible before. "We can see exactly where the connections to the lipids are," says the researcher. The more the scientists learn about photosystem II, the better they understand its precise mode of operation.
However, this method is not only of interest for the photosystem II. "The method is potentially of use for a lot of membrane proteins and will certainly not only work for photosystem II," says Zouni. In this way, many biomolecules could in the future be examined in a more native-like state than before.
Original Paper:
"Native-like Photosystem II Superstructure at 2.44 Å Resolution through Detergent Extraction from the Protein Crystal"; Julia Hellmich, Martin Bommer et al.; Structure, 2014; DOI: 10.1016/j.str.2014.09.007
Research Contacts
Dr. Athina Zouni, Humboldt University Berlin (HU)
Dr. Anja Burkhardt, German Electron Synchrotron of the Helmholtz Association (DESY)
Contact
Dr. Thomas Zoufal
DESY-Press Officer
Tel.: 040 8998-1666
presse(at)desy.de
Hans-Christoph Keller
Office of Press and Public Relations
Humboldt-University Berlin
Tel.: 030 2093-2945
ibou.diop.1(at)hu-berlin.de
